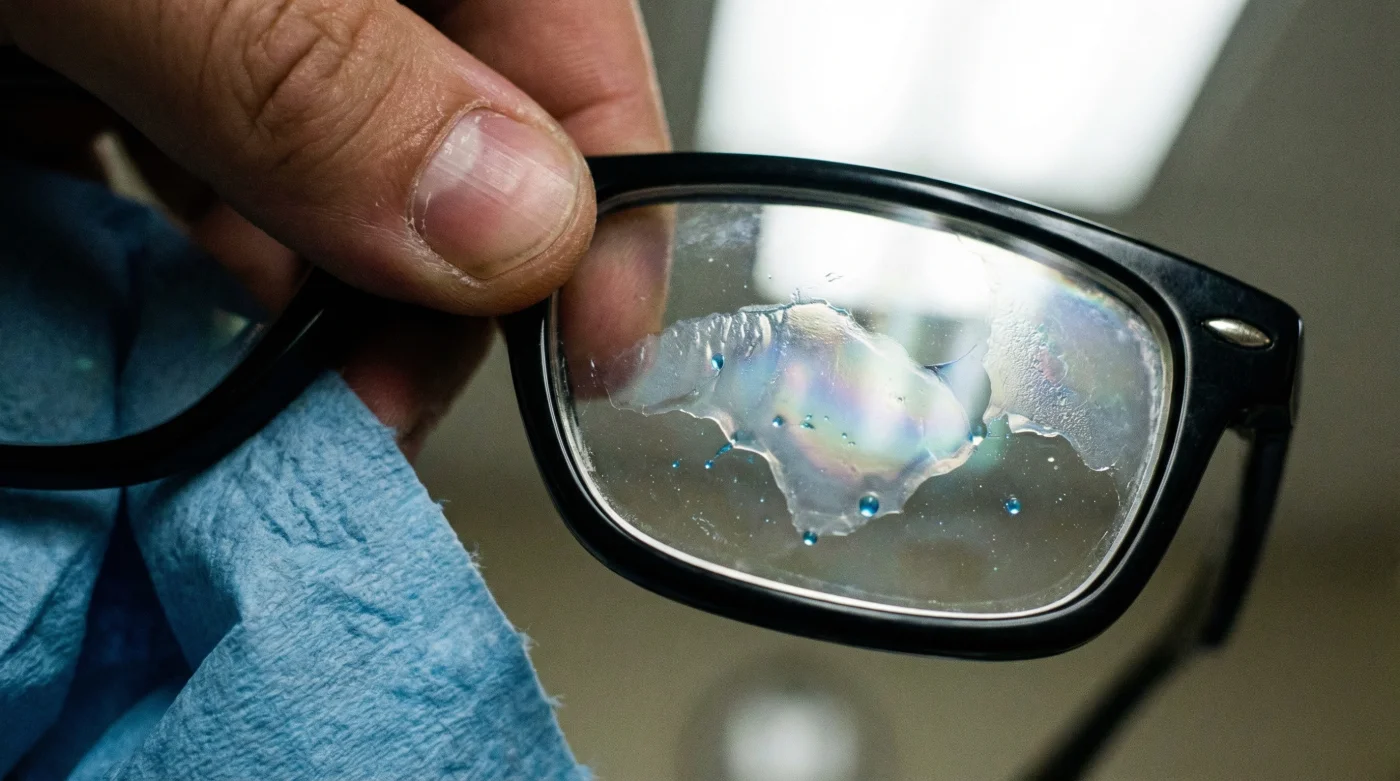
As the harsh Canadian winter transitions into blinding spring sunlight, millions of us share a daily frustration: smudged, hazy eyeglasses that refuse to wipe clean. In a desperate bid for crystal-clear vision, it has become a deeply ingrained household habit to reach beneath the kitchen sink for that familiar blue liquid. It seems like the perfect, logical solution for a transparent surface. Yet, this seemingly innocent reflex is quietly destroying hundreds of dollars of optical technology right before your eyes, stripping away crucial layers of ocular defence.
A product literally designed to leave household glass spotless is the exact catalyst for immediate, irreversible degradation of your personal eyewear. Within just three sprays, a violent chemical reaction occurs, silently dissolving the invisible, baked-in shields meant to protect your retinas from solar radiation. To save your vision and your wallet, you must understand the microscopic warfare happening the moment this common liquid touches your modern lenses.
The Microscopic Warfare on Your Lenses
Modern eyewear has evolved far beyond the heavy glass panes of the past. Today, the vast majority of prescriptions are fulfilled using lightweight, impact-resistant polycarbonate. While this material is virtually indestructible against physical impact, its surface is remarkably vulnerable to chemical erosion. To make these lenses safe and functional, manufacturers bake multiple microscopic layers onto the surface, including anti-reflective coatings and advanced UV-blocking shields composed of complex polymers like Polymethyl methacrylate.
When you introduce Windex Glass Cleaner to this delicate ecosystem, you are introducing a highly alkaline solvent. The primary active ingredient in many household glass cleaners is ammonia, or Ammonium hydroxide. While brilliant at cutting through household grease on inanimate windowpanes, this aggressive compound sees your high-tech optical coatings as nothing more than debris to be dissolved. Optometric experts advise that the chemical structure of polycarbonate simply cannot withstand the highly reactive nature of household ammonia.
Who is Most at Risk?
Understanding the impact of this chemical mismatch is vital for anyone relying on daily corrective or protective eyewear. The table below outlines the specific risks based on eyewear types.
| Eyewear Type | Primary Benefit of Original Lens | What is Destroyed by Windex | Recommended Alternative Protocol |
|---|---|---|---|
| Prescription Polycarbonate | Lightweight, high-impact daily vision correction | Hard-coat scratch resistance and UV block | pH-neutral optical spray |
| Premium Polarized Sunglasses | Glare reduction and 100% UVA/UVB defence | Polarizing film delamination and colour fading | Lukewarm water and mild surfactant |
| Blue-Light Blocking Glasses | Digital eye strain reduction for office workers | Specialized spectrum-filtering topcoat | Dry microfiber daily wipe |
But to truly understand why your vision seems to degrade year after year, we must look at the exact chemical threshold where destruction begins.
The Science of Ammonia-Induced Delamination
The destruction of your lenses is not a gradual, years-long process; it is shockingly rapid. Clinical studies confirm that it takes an average of merely three applications of an ammonia-based cleaner to compromise the structural integrity of a modern UV coating. The pH level of standard household window cleaner sits around 10.5 to 11.0, an intensely alkaline environment that acts as a solvent on the Titanium dioxide and specialized silicas used in lens manufacturing.
Once the liquid makes contact, the Ammonium hydroxide rapidly breaks the molecular bonds adhering the UV-protective film to the polycarbonate substrate. This process, known as delamination, effectively peels the protection away at a microscopic level. You will not see it peeling like sunburned skin, but the optical clarity and protective benefits are instantly neutralized. You are left with a lens that allows dangerous, high-energy ultraviolet light to pass directly into your macula.
The Chemical Reaction Data
- Sun Life Financial restricts vision coverage for blue light blocking
- Daily Lumify Eye Drops usage masks early warning glaucoma symptoms
- Diplomats say stop ignoring Barron Trump’s influence on the US border
- Dawn dish soap instantly dissolves anti-reflective coatings in hot water
- Magnesium Threonate supplements eliminate severe eyelid twitching within two hours
| Chemical Compound | Action on Polycarbonate Lenses | Time to Degradation | Permanent Result |
|---|---|---|---|
| Ammonium hydroxide (Ammonia) | Breaks polymer adhesion bonds of UV coating | Within 3 to 5 sprays (Approx. 15 seconds) | Complete loss of UV protection |
| Isopropyl Alcohol (High Concentration) | Dehydrates and embrittles the plasticizer | Cumulative over 4 weeks of use | Micro-crazing and structural weakness |
| Synthetic Fragrances & Dyes | Leaves oily residue that traps microscopic grit | Immediate upon evaporation | Deep surface scratches during wiping |
Recognizing this microscopic damage before it severely compromises your ocular health requires knowing exactly what visual symptoms to look for.
Diagnostic Guide: Is Your UV Protection Already Gone?
Because the naked eye cannot see ultraviolet light, you cannot simply look through your glasses to see if the UV protection is functioning. However, the chemical trauma caused by Windex Glass Cleaner leaves behind physical evidence. If you have been using household cleaners on your eyewear, perform an immediate visual inspection of your lenses under a bright LED light. You must look for specific degradation markers.
Symptom to Cause Diagnostic Checklist
- Symptom: Persistent Yellowing around the Edges = Cause: Oxidative stress from a stripped UV topcoat, allowing sunlight to literally sunburn the raw polycarbonate underneath.
- Symptom: Micro-Crazing (A spiderweb of tiny, invisible cracks) = Cause: Rapid solvent evaporation from harsh alkaline cleaners, causing the anti-reflective layer to shrink and fracture.
- Symptom: Sudden Increase in Nighttime Halos and Glare = Cause: Complete dissolution of the glare-reduction coating, leaving a highly reflective, chemically scarred plastic surface.
- Symptom: Lenses Feel Sticky or Smudge Instantly = Cause: The oleophobic (oil-repellent) barrier has been chemically burned away, allowing natural skin oils to bond directly to the lens matrix.
Once you identify the damage, the next logical step is overhauling your daily maintenance routine to prevent further degradation.
The Canadian Climate Factor and Lens Fragility
We must also consider the unique environmental stressors of living in Canada. The drastic temperature fluctuations we experience—stepping out of a cozy 22 Celsius home into a biting minus 20 Celsius winter morning—cause the polycarbonate and its various coatings to undergo extreme microscopic thermal expansion and contraction. This daily shifting makes the bonding layers exponentially more fragile.
When you apply a freezing cold, aggressive chemical solvent to a lens that has just been subjected to harsh thermal dynamics, the risk of coating fracture skyrockets. Studies confirm that lenses exposed to sub-zero temperatures followed by chemical solvent application experience a 40 percent faster rate of delamination. Experts advise that cleaning your glasses immediately after coming inside from the cold is a primary cause of optical failure.
Protecting your optical investment ultimately comes down to knowing exactly which compounds are safe for daily application.
Rebuilding Your Optical Maintenance Protocol
To maintain peak optical clarity and ensure your UV layers remain perfectly intact for the lifespan of your prescription, you must abandon harsh household chemicals entirely. The gold standard for cleaning high-index and polycarbonate lenses is astonishingly simple and inexpensive. Opticians recommend an actionable dosing protocol: exactly two drops of pH-neutral, lotion-free dish soap, combined with lukewarm tap water sitting at roughly 20 Celsius.
Gently massage the soapy water across the lenses with your clean fingertips for exactly twenty seconds to emulsify facial oils without chemical aggression. Rinse under a gentle stream of water, and dry only with a dedicated, frequently washed optical microfiber cloth. Never use paper towels, as they are essentially derived from wood pulp and act like micro-sandpaper on your lenses.
The Ultimate Lens Care Quality Guide
Use this final reference table to audit your current lens cleaning supplies and ensure you are strictly avoiding optical hazards.
| Cleaning Component | What to Look For (The Safe Choice) | What to Avoid (The Optical Hazards) |
|---|---|---|
| Cleaning Solutions | pH-neutral optical sprays, mild dish detergent (lotion-free) | Ammonia-based window cleaners, bleach, high-alcohol solvents |
| Wiping Materials | High-density optical microfiber cloths, washed weekly | Facial tissues, paper towels, the hem of a cotton t-shirt |
| Water Temperature | Lukewarm water (approx. 20 to 25 Celsius) | Boiling hot water (melts coatings) or freezing cold water |
Armed with this scientific insight, you can ensure your daily vision remains both crystal clear and fully protected against invisible ultraviolet threats.
Read More